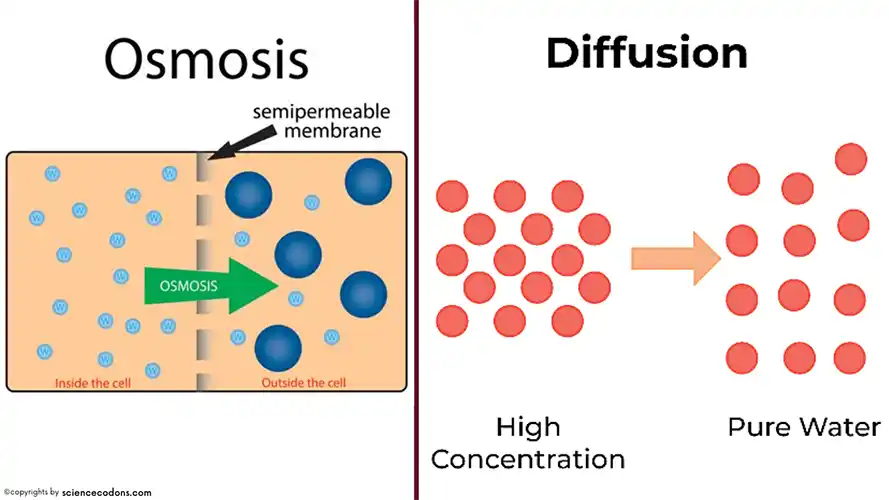
In the complex world of biological processes, osmosis and simple diffusion are two fundamental mechanisms that play distinct roles in maintaining equilibrium within living organisms. While both processes have a lot in common, they differ in their driving forces and the types of substances. In this article, we talk about the main differences between osmosis and simple diffusion.
main differences between osmosis and simple diffusion
| Attributes | Osmosis | Simple diffusion |
|---|---|---|
| Direction | Water molecules move from an area with a higher concentration of water to an area with a lower concentration. | Movement of substances from a higher concentration area to a lower concentration area. |
| Type of molecules involved | Only water molecules | Solutes(salts) such as ions or any solid, liquid or gas molecule |
| Membrane | Only Semipermeable membrane | All types |
| Energy | the potential energy of water. | The kinetic energy of substances |
| Main force | Osmotic pressure | Difference in concentration gradient |
| Time | Slower than Simple diffusion | Faster than osmosis |
Other differences and similarities
Diffusion and osmosis both occur through passive transport. This means that no additional energy is required to conduct them. They simply involve the movement of particles from a higher concentration to a lower concentration.
One of the major differences between these two types is the environment in which these processes take place. This means diffusion occurs in any medium( solid, liquid, or gas) while osmosis can only occur in a liquid medium. The purpose of diffusion is to reach a balance in the energy concentration in the biological system. On the other hand, osmosis requires the balance of solute concentration between the intracellular environment and its external environment.